Is using theory and conceptual frameworks in studies just “pie-in-the-sky” stuff? Do they have any practical use? Or are they merely for academics in ivory towers?
This blog about theory-testing research1 may affect your answers.
What is it? At its most basic, theory or framework is a set of statements that describe part of reality. Those related statements (called propositions) outline the relationship between two or more ideas (called concepts). One example of a set of propositions is: “Work stress leads to burnout; burnout leads to poor work outcomes; mindfulness practice leads to lower burnout and thus to better work outcomes.” These statements describe the relationships between concepts of “work stress,” “burnout,” “poor work outcomes,” and “mindfulness practice.”
Each concept has 1) an abstract dictionary-type, conceptual definition & 2) a concrete, measurable, operational definition. For example, Maslach conceptually defined burnout as a combination of emotional exhaustion, depersonalization, and lower personal accomplishment; then those concepts are operationally defined as a self-reported burnout score on Maslach’s Burnout Inventory (MBI).
Some theories are named for their authors–like Einstein’s theory of relativity expressed in a single proposition about the relationship between concepts of energy, mass, & speed of light. Einstein’s theory & propositions of other theory/frameworks describe our existing knowledge about a topic based on evidence and logical connections.
To connect your study with such existing knowledge, take these steps:
1) Identify a theory/framework that conceptually & operationally defines your concept of interest and states its relationship to other concepts. Start by looking in the library for articles on your topic.
2) Accept most of the theory/framework’s propositions as true without testing them yourself (called assumptions). All studies assume a lot to be true already–meaning they have a lot of assumptions. It’s the way science works because you can’t test everything at once.
3) Identify a proposition that you want to test, and write it in a testable form as a hypotheses or research questions. You will be testing only a tiny piece of the theory/framework, perhaps by examining the concepts in a new setting, with new methods, or in a different or larger sample. For example, you might want to test an intervention to see if it reduces burnout (e.g., Hypothesis: “ICU staff using a mindfulness phone app will report lower burnout than those who do not use the app.”)
4) When your study is complete, discuss how your findings confirm or disconfirm the theory/framework. Your logic and research are now a part of what we know (or think we know).
Conclusion: Of course there’s much more that could be said on this topic. Let me know what to add in the comments. -Dr.H
Questions for thot:
So, do you think theory/conceptual frameworks are just “pie in the sky” without practical value? If so, how would you build a study on existing knowledge? If you think they ARE practical, how would you use them to study your topic of interest? Explain how you have or have not used propositions in a study.
- Theory-building research is a different inductive path. Theory-testing is more deductive. ↩︎


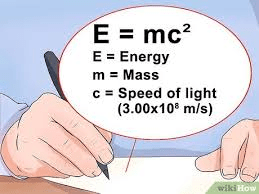







 In a quasi experimental design
In a quasi experimental design


