 Quasi-experiments are a lot of work, yet don’t have the same scientific power to show cause and effect, as do randomized controlled trials (RCTs). An RCT would provide better support for any hypothesis that X causes Y. [As a quick review of what quasi-experimental versus RCT studies are, see “Of Mice & Cheese” and/or “Out of Control (Groups).”]
Quasi-experiments are a lot of work, yet don’t have the same scientific power to show cause and effect, as do randomized controlled trials (RCTs). An RCT would provide better support for any hypothesis that X causes Y. [As a quick review of what quasi-experimental versus RCT studies are, see “Of Mice & Cheese” and/or “Out of Control (Groups).”]
So why do quasi-experimental studies at all? Why not always do RCTs when we are testing cause and effect? Here are 3 reasons:
#1 Sometimes ETHICALLY the researcher canNOT randomly assign subjects to a control  and an experimental group. If the researcher wants to compare health outcomes of smokers with non-smokers, the researcher cannot assign some people to smoke and others not to smoke! Why? Because we already know that smoking has significant harmful effects. (Of course, in a dictatorship, by using the police a researcher could assign them to smoke or not smoke, but I don’t think we wanna go there.)
and an experimental group. If the researcher wants to compare health outcomes of smokers with non-smokers, the researcher cannot assign some people to smoke and others not to smoke! Why? Because we already know that smoking has significant harmful effects. (Of course, in a dictatorship, by using the police a researcher could assign them to smoke or not smoke, but I don’t think we wanna go there.)
#2 Sometimes PHYSICALLY the researcher canNOT randomly assign subjects to control &  experimental groups. If the researcher wants to compare health outcomes of
experimental groups. If the researcher wants to compare health outcomes of
individuals from different countries, it is physically impossible to assign country of origin.
#3 Sometimes FINANCIALLY the researcher canNOT afford to assign subjects randomly  to control & experimental groups. It costs $ & time to get a list of subjects and then assign them to control & experimental groups using random numbers table or drawing names from a hat.
to control & experimental groups. It costs $ & time to get a list of subjects and then assign them to control & experimental groups using random numbers table or drawing names from a hat.
Thus, researchers sometimes are left with little alternative, but to do a quasi-experiment as the next best thing to an RCT, then discuss its limitations in research reports.
Critical Thinking: You read a research study in which a researcher recruits the 1st 100 patients on a surgical ward January-March quarter as a control group. Then the researcher recruits the 2nd 100 patients on that same surgical ward April-June for the experimental group. With the experimental group, the staff uses a new, standardized pain script for better pain communications. Then the pain communication outcomes of each group are compared statistically.
- Is this a quasi-experiment or a randomized controlled trial (RCT)?
- What factors (variables) might be the same among control & experimental groups in this study?
- What factors (variables) might be different between control & experimental groups that might affect study outcomes?
- How could you design an ethical & possible RCT that would overcome the problems with this study?
- Why might you choose to do the study the same way that this researcher did?
For more info: see “Of Mice & Cheese” and/or “Out of Control (Groups).”






 improve the homemade masks?
improve the homemade masks? 



 case study, systematic review, or clinical practice guideline? Check out these standardized reporting guidelines:
case study, systematic review, or clinical practice guideline? Check out these standardized reporting guidelines: 
 Quasi-experiments are a lot of work, yet don’t have the same scientific power to show cause and effect, as do randomized controlled trials (RCTs). An RCT would provide better support for any hypothesis that X causes Y. [As a quick review of what quasi-experimental versus RCT studies are, see
Quasi-experiments are a lot of work, yet don’t have the same scientific power to show cause and effect, as do randomized controlled trials (RCTs). An RCT would provide better support for any hypothesis that X causes Y. [As a quick review of what quasi-experimental versus RCT studies are, see  and an experimental group. If the researcher wants to compare health outcomes of smokers with non-smokers, the researcher cannot assign some people to smoke and others not to smoke! Why? Because we already know that smoking has significant harmful effects. (Of course, in a dictatorship, by using the police a researcher could assign them to smoke or not smoke, but I don’t think we wanna go there.)
and an experimental group. If the researcher wants to compare health outcomes of smokers with non-smokers, the researcher cannot assign some people to smoke and others not to smoke! Why? Because we already know that smoking has significant harmful effects. (Of course, in a dictatorship, by using the police a researcher could assign them to smoke or not smoke, but I don’t think we wanna go there.) experimental groups. If the researcher wants to compare health outcomes of
experimental groups. If the researcher wants to compare health outcomes of to control & experimental groups. It costs $ & time to get a list of subjects and then assign them to control & experimental groups using random numbers table or drawing names from a hat.
to control & experimental groups. It costs $ & time to get a list of subjects and then assign them to control & experimental groups using random numbers table or drawing names from a hat.
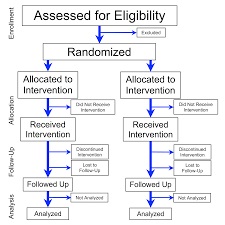
 In a quasi experimental design
In a quasi experimental design





 perimental drug X will have better cardiac function than will heart failure patients who receive standard drug Y.” You can see that the researcher is manipulating the drug (independent variable) that patients will receive. And patient cardiac outcomes are expected to vary—in fact cardiac function is expected to be better—for patients who receive the experimental drug X.
perimental drug X will have better cardiac function than will heart failure patients who receive standard drug Y.” You can see that the researcher is manipulating the drug (independent variable) that patients will receive. And patient cardiac outcomes are expected to vary—in fact cardiac function is expected to be better—for patients who receive the experimental drug X. 1st – Identify the population in the hypothesis—the population does not vary (& so, it is not a variable). 2nd – Identify the independent variable–This will be the one that is the cause & it will vary. 3rd – Identify the dependent variable–This will be the one that is the outcome & its variation depends on changes/variation in the independent variable.
1st – Identify the population in the hypothesis—the population does not vary (& so, it is not a variable). 2nd – Identify the independent variable–This will be the one that is the cause & it will vary. 3rd – Identify the dependent variable–This will be the one that is the outcome & its variation depends on changes/variation in the independent variable.